Torium
90
Th
Kumpulan
Tiada
Kala
7
Blok
f
Proton
Elektron
Neutron
90
90
142
Ciri-Ciri Am
Nombor atom
90
Berat atom
232.03806
Nombor jisim
232
Kategori
Aktinid
Warna
Perak
Radioaktif
Ya
Named after Thor, Scandinavian god of war
Struktur hablur
Pusat Wajah Kubik
Sejarah
Thorium was discovered by Jöns Jacob Berzelius in 1828, in Stockholm, Sweden.
Thorium was first observed to be radioactive in 1898, independently, by Polish-French physicist Marie Curie and German chemist Gerhard Carl Schmidt.
The crystal bar process was discovered by Anton Eduard van Arkel and Jan Hendrik de Boer in 1925 to produce high-purity metallic thorium.
Thorium was first observed to be radioactive in 1898, independently, by Polish-French physicist Marie Curie and German chemist Gerhard Carl Schmidt.
The crystal bar process was discovered by Anton Eduard van Arkel and Jan Hendrik de Boer in 1925 to produce high-purity metallic thorium.
Bilangan elektron per petala
2, 8, 18, 32, 18, 10, 2
Konfigurasi elektron
[Rn] 6d2 7s2
Thorium fuel research is continuing in several countries including the USA and India
Ciri-Ciri Fizikal
Fasa
Pepejal
Ketumpatan
11.7 g/cm3
Takat lebur
2115.15 K | 1842 °C | 3347.6 °F
Takat didih
5061.15 K | 4788 °C | 8650.4 °F
Haba pelakuran
16 kJ/mol
Haba pengewapan
530 kJ/mol
Muatan haba molar
0.113 J/g·K
Banyak pada kerak bumi
0.0006%
Banyak pada alam semesta
4×10-8%
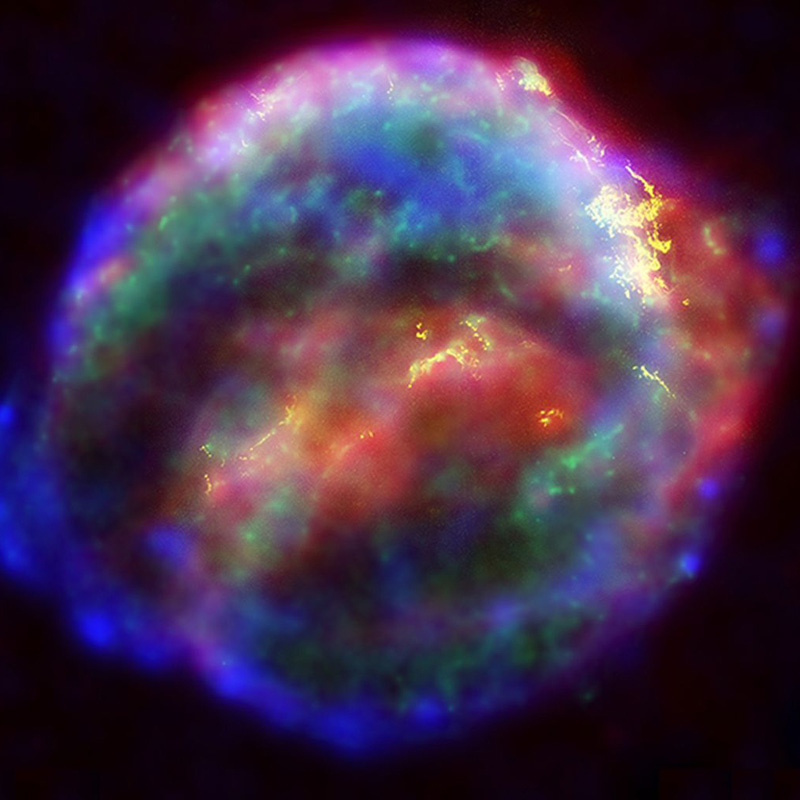
Kredit imej: Wikimedia Commons (NASA/ESA/JHU/R.Sankrit & W.Blair)
The Earth's thorium originated in the death throes of ancient stars
Nombor CAS
7440-29-1
Nombor PubChem CID
23960
Ciri-Ciri Atom
Jejari atom
179 pm
Jejari kovalen
206 pm
Keelektronegatifan
1.3 (Skala Pauling)
Kebolehan mengion
6.3067 eV
Isipadu atom
19.9 cm3/mol
Daya pengaliran terma
0.54 W/cm·K
Keadaan pengoksidaan
2, 3, 4
Aplikasi
Thorium is used to coat tungsten filaments in light bulbs.
It is also used in its oxide form in gas tungsten arc welding to increase the high-temperature strength of tungsten electrodes and improve arc stability.
Thorium-magnesium alloys are used in the aerospace industry for aircraft engines.
It is also used in its oxide form in gas tungsten arc welding to increase the high-temperature strength of tungsten electrodes and improve arc stability.
Thorium-magnesium alloys are used in the aerospace industry for aircraft engines.
Thorium is highly radioactive
Isotop
Isotop stabil
232ThIsotop tidak stabil
209Th, 210Th, 211Th, 212Th, 213Th, 214Th, 215Th, 216Th, 217Th, 218Th, 219Th, 220Th, 221Th, 222Th, 223Th, 224Th, 225Th, 226Th, 227Th, 228Th, 229Th, 230Th, 231Th, 233Th, 234Th, 235Th, 236Th, 237Th, 238Th